Your consumer products news reporter
Provided by AGPGlobal Plasmid DNA Manufacturing Market Anticipates Impressive Growth Trajectory at a CAGR of ~18% by 2034 | DelveInsight
The plasmid DNA manufacturing market is witnessing strong growth driven by rising demand for gene therapies, DNA vaccines, and cell-based therapies such as CAR-T treatments. Increasing clinical trials in genetic medicine and expanding applications in mRNA vaccine production are further accelerating the need for high-quality plasmid DNA. Biopharmaceutical companies are investing heavily in scalable and GMP-compliant manufacturing capabilities to meet growing demand.
New York, USA, May 04, 2026 (GLOBE NEWSWIRE) -- Global Plasmid DNA Manufacturing Market Anticipates Impressive Growth Trajectory at a CAGR of ~18% by 2034 | DelveInsight
The plasmid DNA manufacturing market is witnessing strong growth driven by rising demand for gene therapies, DNA vaccines, and cell-based therapies such as CAR-T treatments. Increasing clinical trials in genetic medicine and expanding applications in mRNA vaccine production are further accelerating the need for high-quality plasmid DNA. Biopharmaceutical companies are investing heavily in scalable and GMP-compliant manufacturing capabilities to meet growing demand.
DelveInsight’s Plasmid DNA Manufacturing Market Insights report provides the current and forecast market analysis, individual leading plasmid DNA manufacturing companies’ market shares, challenges, plasmid DNA manufacturing market drivers, barriers, trends, and key plasmid DNA manufacturing companies in the market.
Plasmid DNA Manufacturing Market Summary
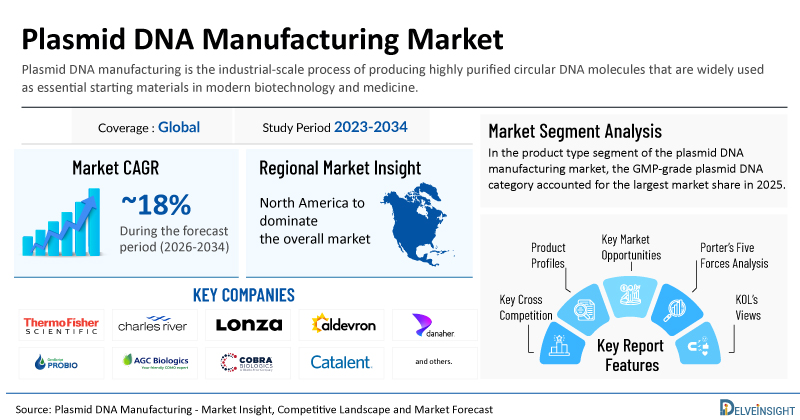
- 2025 Plasmid DNA Manufacturing Market Size: USD 2.4 Billion
- 2034 Projected Plasmid DNA Manufacturing Market Size: USD 10.8 Billion
- Plasmid DNA Manufacturing Market Growth Rate (2026-2034): ~18%
- Largest Plasmid DNA Manufacturing Market: North America
- Largest Product Type Segment: GMP-grade Plasmid DNA Category
- Key Companies in the Plasmid DNA Manufacturing Market: Thermo Fisher Scientific, Charles River Laboratories, Lonza, Aldevron (Danaher), GenScript ProBio, AGC Biologics, Cobra Biologics (Charles River), Catalent, WuXi AppTec, WuXi Advanced Therapies, Eurogentec, Sartorius (BIA Separations), ProBio, PharmaCell, Pharmaron, Esco Aster, AGTC (Applied Genetic Technologies Corp.), VGXI (GeneOne Life Science), PlasmidFactory, VectorBuilder, and others
To read more about the latest highlights related to the plasmid DNA manufacturing market, get a snapshot of the key highlights @ https://www.delveinsight.com/sample-request/plasmid-dna-manufacturing-market
Key Factors Contributing to the Rise in Growth of the Plasmid DNA Manufacturing Market
- Rising Demand for Gene and Cell Therapies: Plasmid DNA is a critical raw material used in the production of viral vectors and non-viral delivery systems for gene and cell therapies. As more therapies progress through clinical trials and commercialization, the need for high-quality plasmid DNA continues to increase.
- Expansion of mRNA and DNA Vaccine Development: The success of mRNA vaccines accelerated demand for plasmid DNA, which serves as a template in mRNA manufacturing. Additionally, growing interest in DNA vaccines for infectious diseases and oncology is creating new growth opportunities for plasmid DNA producers.
- Increasing Outsourcing to CDMOs: Many biotech and pharmaceutical companies lack in-house GMP-grade plasmid manufacturing capabilities. As a result, they are increasingly partnering with specialized contract development and manufacturing organizations (CDMOs), boosting outsourced production demand.
- Growing Pipeline of Genetic Medicines: The expanding pipeline of therapies targeting rare diseases, inherited disorders, cancer, and cardiovascular conditions is significantly increasing demand for plasmid DNA during preclinical, clinical, and commercial stages.
- Technological Advancements in Manufacturing: Innovations such as high-yield fermentation systems, improved purification technologies, automation, and closed-system manufacturing are enhancing productivity, scalability, and product quality. These advancements make large-scale production more feasible and cost-effective.
- Increasing Need for GMP-Grade Material: Regulatory agencies require highly pure and consistent plasmid DNA for therapeutic applications. This has led to rising investments in GMP-compliant facilities, quality control systems, and advanced bioprocessing infrastructure.
- Strategic Investments and Capacity Expansion: Leading manufacturers are expanding global production footprints through facility expansions, mergers, acquisitions, and strategic collaborations to meet rising demand and reduce supply bottlenecks.
- Increasing Focus on Personalized Medicine: The shift toward precision medicine and patient-specific therapies is driving demand for customized plasmid DNA constructs, creating opportunities for flexible and niche manufacturing services.
Get a sneak peek at the plasmid DNA manufacturing market dynamics @ Plasmid DNA Manufacturing Market Trends
Regional Plasmid DNA Manufacturing Market Insights
North America
- North America held the largest share of the global plasmid DNA manufacturing market in 2025, representing 44.12% of total regional revenue.
- The region’s leadership is driven by the strong presence of major biotechnology and pharmaceutical companies, highly developed biomanufacturing capabilities, and a dense network of CDMOs focused on gene and cell therapy manufacturing.
- It also benefits from substantial investments in research and development, growing adoption of advanced therapies such as gene therapies, CAR-T treatments, and mRNA vaccines, along with a favorable regulatory environment supported by agencies such as the U.S. Food and Drug Administration.
- Furthermore, the rising number of clinical trials and increasing commercialization of advanced biologics continue to reinforce North America’s dominant position in the global plasmid DNA manufacturing market.
Europe
- The European plasmid DNA manufacturing market is witnessing strong expansion, driven by the region’s well-established biotechnology landscape, rising investments in gene and cell therapy programs, and increasing need for GMP-grade plasmid DNA for both clinical and commercial use.
- Countries including Germany, the U.K., and France are becoming major centers of activity, supported by sophisticated research infrastructure, supportive regulatory environments, and expanding CDMO capacities.
- Growth is further fueled by the growing number of clinical trials and broader use of plasmid DNA in gene therapies, DNA vaccines, and immunotherapy applications.
- In addition, ongoing innovations in plasmid production technologies, such as automation and high-efficiency fermentation platforms, are enhancing scalability and operational efficiency across European manufacturing sites.
- Recent developments continue to reinforce this positive market outlook.
Asia-Pacific
- The Asia Pacific (APAC) region is becoming a significant growth engine for the plasmid DNA manufacturing market, driven by expanding biotechnology capabilities, rising investments in gene and cell therapy research, and the increasing presence of contract development and manufacturing organizations (CDMOs) in countries such as China, Japan, South Korea, and India.
- Demand for plasmid DNA is growing strongly as it serves as a critical raw material for gene therapies, DNA vaccines, and mRNA-based therapeutics, supported by the increasing prevalence of chronic and genetic disorders as well as a rapidly aging population.
- Furthermore, substantial investments from governments and private sector players in domestic biomanufacturing infrastructure are boosting clinical trial activity and advancing the commercialization of next-generation therapies.
To know more about why North America is leading the market growth in the plasmid DNA manufacturing market, get a snapshot of the Plasmid DNA Manufacturing Market Share
Recent Developmental Activities in the Plasmid DNA Manufacturing Market
- In December 2025, Aldevron announced the continued expansion of GMP plasmid DNA manufacturing capabilities to support growing gene and cell therapy demand.
- In November 2025, Bharat Biotech announced the launch of its new subsidiary, “Nucelion Therapeutics,” which includes a GMP-compliant facility capable of producing plasmids and viral vectors to support cell and gene therapy development.
- In April 2025, Thermo Fisher Scientific expanded its plasmid DNA manufacturing capacity in the United States, introducing enhanced single-use bioreactors and automated purification systems to support large-scale production for gene therapy and mRNA vaccine applications. This expansion was aimed at addressing growing demand from clinical and commercial gene therapy pipelines.
What is Plasmid DNA Manufacturing?
Plasmid DNA manufacturing is the industrial-scale process of producing highly purified circular DNA molecules (plasmids) that are widely used as essential starting materials in modern biotechnology and medicine. These plasmids serve as critical components in gene therapies, DNA vaccines, cell therapies such as CAR-T cell therapy, and viral vector production for AAV and lentiviral therapies. The manufacturing process typically begins with designing and cloning the desired genetic sequence into a bacterial host, most commonly Escherichia coli, followed by fermentation to amplify the plasmid DNA. After cell growth, the bacteria are harvested and lysed to release the plasmids, which are then purified through multiple downstream processing steps such as filtration and chromatography to remove impurities, including genomic DNA, RNA, proteins, and endotoxins. Final products undergo stringent quality control testing to ensure identity, purity, potency, and regulatory compliance under Good Manufacturing Practice (GMP) standards. As demand for advanced therapies grows, plasmid DNA manufacturing has become a strategic backbone of the biopharmaceutical supply chain.
| Plasmid DNA Manufacturing Market Report Metrics | Details |
| Coverage | Global |
| Study Period | 2023–2034 |
| Plasmid DNA Manufacturing Market CAGR | ~18% |
| Plasmid DNA Manufacturing Market Size by 2034 | USD 10.8 Billion |
| Key Plasmid DNA Manufacturing Companies | Thermo Fisher Scientific, Charles River Laboratories, Lonza, Aldevron (Danaher), GenScript ProBio, AGC Biologics, Cobra Biologics (Charles River), Catalent, WuXi AppTec, WuXi Advanced Therapies, Eurogentec, Sartorius (BIA Separations), ProBio, PharmaCell, Pharmaron, Esco Aster, AGTC (Applied Genetic Technologies Corp.), VGXI (GeneOne Life Science), PlasmidFactory, VectorBuilder, and others |
Plasmid DNA Manufacturing Market Assessment
-
Plasmid DNA Manufacturing Market Segmentation
- Plasmid DNA Manufacturing Market Segmentation By Product Type: GMP-grade Plasmid DNA and Research-grade Plasmid DNA
- Plasmid DNA Manufacturing Market Segmentation By Application: Cell Therapy Manufacturing, Gene Therapy Manufacturing, DNA/RNA Vaccine Development, Viral Vector Manufacturing, and Others
- Plasmid DNA Manufacturing Market Segmentation By Production Scale: Commercial, Clinical, and Preclinical
- Plasmid DNA Manufacturing Market Segmentation By End User: Pharmaceutical & Biotechnology Companies, Contract Development & Manufacturing Organizations (CDMOs), and Others
- Plasmid DNA Manufacturing Market Segmentation By Geography: North America, Europe, Asia-Pacific, and Rest of World
- Porter’s Five Forces Analysis, Product Profiles, Case Studies, KOL’s Views, Analyst’s View
Which MedTech key players in the plasmid DNA manufacturing market are set to emerge as the trendsetter explore @ Plasmid DNA Manufacturing Market Analysis
Table of Contents
| 1 | Plasmid DNA Manufacturing Market Report Introduction |
| 2 | Plasmid DNA Manufacturing Market Executive Summary |
| 3 | Plasmid DNA Manufacturing Market Key Factors Analysis |
| 4 | Impact Analysis |
| 5 | Regulatory Analysis |
| 6 | Plasmid DNA Manufacturing Market Porter’s Five Forces Analysis |
| 7 | Plasmid DNA Manufacturing Market Assessment |
| 8 | Competitive Landscape |
| 9 | Startup Funding & Investment Trends |
| 10 | Plasmid DNA Manufacturing Market Company and Product Profiles |
| 11 | KOL Views |
| 12 | Project Approach |
| 13 | About DelveInsight |
| 14 | Disclaimer & Contact Us |
Interested in knowing the plasmid DNA manufacturing market share by 2034? Click to get a snapshot of the Plasmid DNA Manufacturing Market Size
Related Reports
AAV Vector Manufacturing Market
AAV Vector Manufacturing Market Insights, Competitive Landscape and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key AAV vector manufacturing companies, including Thermo Fisher Scientific, Catalent Gene Therapy, Lonza Group, Oxford Biomedica, FUJIFILM Diosynth Biotechnologies, WuXi Advanced Therapies, Charles River Laboratories, SignaGen Laboratories, Batavia Biosciences, Andelyn Biosciences, Forge Biologics, Viralgen Vector Core, SK Pharmteco, Aldevro, Polyplus, Avirmax CMC, AGC Biologics, VectorBuilder, PackGene Biotech, Vector Biolabs, and others.
Cell and Gene Therapy Manufacturing Market
Cell and Gene Therapy Manufacturing Market Insight, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key cell and gene therapy manufacturing companies, including Lonza, Catalent, Inc., Thermo Fisher Scientific, Inc., Charles River Laboratories, WuXi AppTec, Merck KGaA, Takara Bio Inc., Oxford Biomedica PLC, Genezen, FUJIFILM Holdings Corporation, Nikon Corporation, The Discovery Labs LLC, RoslinCT, JRS Pharma, FinVector, Sarepta Therapeutics, Inc., BioCentriq, Andelyn Biosciences, ElevateBio, Anemocyte Srl, and others.
mRNA Synthesis and Manufacturing Market
mRNA Synthesis and Manufacturing Market Insight, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key mRNA synthesis and manufacturing companies, including Thermo Fisher Scientific Inc., New England Biolabs, TriLink BioTechnologies, DH Life Sciences, LLC., F. Hoffmann-La Roche Ltd., Telesis Bio Inc., Promega Corporation, Jena Bioscience, GenScript, RiboPro, Takara Bio, Lonza, Sigma-Aldrich, Agilent Technologies, Integrated DNA Technologies, Inc., and others.
Viral Vector Manufacturing Market
Viral Vector Manufacturing Market Insight, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key viral vector manufacturing companies, including Oxford Biomedica, Thermo Fisher Scientific Inc., Charles River Laboratories, FUJIFILM Diosynth Biotechnologies, Batavia Biosciences, CEVEC Pharmaceuticals GmbH, REGENXBIO Inc., BioNTech IMFS, AGC Biologics, ANDELYN BIOSCIENCES, INSTITUT MERIEUX, IDT Biologika, Lonza, Genezen, NecstGen, Avid Bioservices, Inc., Takara Bio Inc., VIROCELL BIOLOGICS, LTD., and others.
Vaccine Contract Manufacturing Market
Vaccine Contract Manufacturing Market Insight, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key vaccine contract manufacturing companies, including Sanofi, Biovac, Emergent BioSolutions Inc., Lonza Group, Catalent Inc., Boehringer Ingelheim BioXcellence, Wockhardt, WuXi Biologics, Moderna, Inc., Syngene International, Meridian Life Sciences, Eurogentec, IDT Biologika, Serum Institute of India, GlaxoSmithKline (GSK), Soligenix Inc., Curia, Cobra Biologics, BioReliance, Recipharm, Jubilant HollisterStier, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.

Contact Us Shruti Thakur info@delveinsight.com +14699457679
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
